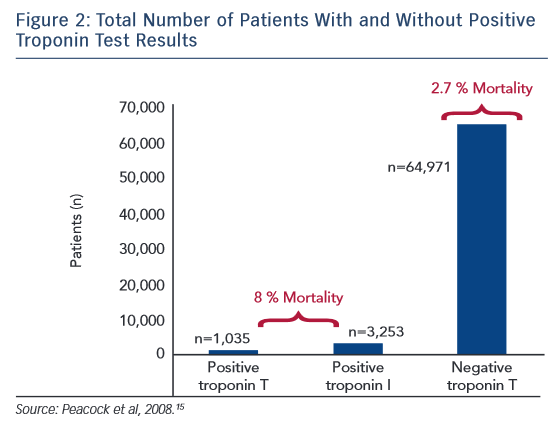
Furthermore, it mandates the logistics between physician and laboratory. While immunoassays are highly developed and accurate, extensive sample preparation including cumbersome blood 7 draws are required. The state-of-the-art 4 troponin quantitation mechanism is based on immunoassays 5 involving the use of two or more antibodies, one of which is labeled, typically with a chemiluminescent tag, which adds another level of complexity 6 in the analysis. Therefore, the development of new technologies that can allow early non-invasive detection of myocardial injury is imperative.ĭetection of cardiac troponins 3 to assess cardiac injury has been around since the 1990s. Furthermore, 1 out of 5 myocardial infarctions (MI) is asymptomatic (silent), leading to nearly 200,000 silent MIs each year in the US 2. Over 80% of these are due to non-cardiac causes, resulting in an unnecessary burden in the ED, revealing the need for an instant non-invasive screening technique that can streamline the ED workflows 1. Over 10 million patients present with chest pain 1 in emergency departments (ED) in the United States alone. Further, potential pitfalls associated with infrared spectroscopic mode of inquiry are outlined including requisite steps needed for improving the precision and overall diagnostic value of the device in future studies. This preliminary work introduces the potential of a bloodless transdermal measurement of troponin-I based on molecular spectroscopy. We demonstrate a significant correlation ( r = 0.7774, P < 0.001, n = 52 biologically independent samples) between optically-derived data and blood-based immunoassay measurements with and an area under receiver operator characteristics of 0.895, sensitivity of 96.3%, and specificity of 60% for predicting a clinically meaningful threshold for defining elevated Troponin I. Patients were recruited with suspected acute coronary syndrome. Our device relies on infrared spectroscopic detection of troponin-I through the dermis and is tested in stepwise laboratory, benchtop, and clinical studies. In this communication, we present the development and early validation of non-invasive transdermal monitoring of cardiac troponin-I to detect its elevated state. This modality further restricts the number of measurements given the clinical context of the patient. Current standard-of-care troponin assays provide a snapshot or momentary view of the levels due to the requirement of a blood draw. 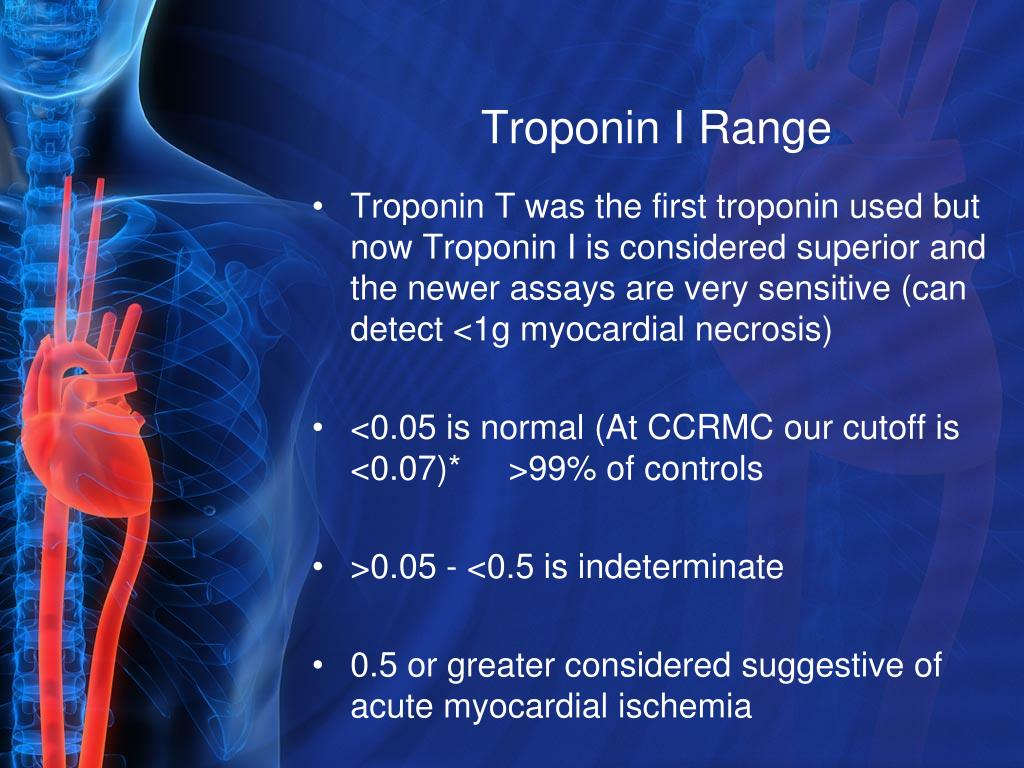
For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics.The levels of circulating troponin are principally required in addition to electrocardiograms for the effective diagnosis of acute coronary syndrome. Check with your local representative for availability in specific markets. 
Not all products are available in all regions. GDPR Statement | Declaration for California Compliance Law. 
Any person depicted in such photographs is a model. Photos displayed are for illustrative purposes only. Your use of this website and the information contained herein is subject to our Website Terms and Conditions and Privacy Policy. The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. This website is governed by applicable U.S. No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the company. Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
